���������ĺϳɼ��俹������Ԣ�1
���ߣ������ ����� ��͢ɭ �Ŵ�־ ������ ������
��λ���ڶ���ҽ��ѧҩѧԺ���Ϻ�200433
�ؼ��ʣ������컷����������������������
ժ Ҫ Ϊ��Ѱ������Ч�Ͷ��Ŀ����ҩժ Ҫ Ϊ��Ѱ������Ч�Ͷ��Ŀ����ҩ�����ݵ�������ҩ��Ĺ�Ч��ϵ�����û�������ƺϳ���21��2��2-˫ȡ��-4-(2��4-��������)-4-(1H-1��2��4-����-1-��)��-1��3�����컷������ṹ��Ԫ�ط�������˴Ź�����֤ʵ.���������⿹�������������������е�Ŀ�껯�����6�ֳ������²�������в�ͬ�̶ȵĿ�������ԣ���dz�������Ч��Ҫ�ȶ�������Ч���ã����л�����(��7)��(��12)��(��13)��(��15)�Ļ������ù���ͪ�����൱��������(��12)�������������������к�ǿ����������.
, http://www.100md.com
Synthesis and Antifungal Activity of Triazole Derivatives ��
Wu Yijie,Yang Jiqiu,Zhou Tingsen,Zhang Dazhi,Liu Chaomei,Zhou Mingde
(College of Pharmacy,Second Military Medical University,Shanghai 200433 )
Abstract In an effort to search for more potent and less toxi c antifungal agents,according to the structure-activity relationships and antin ycotic mechanism of the azole compounds,we designed and synthesized twenty-one 2��2-disu bstituted-4-(2��4-difluorophenyl)-4-(1H-1��2��4-triazol-1-yl)methyl -1,3-dioxolane.Their structures were confirmed by 1H-NMR and elementar y analysis.Preliminary results of the antifungal tests in vitro showed that all compoun d s exhibited activity against the common pathogenic fungi.Generally,the title com pounds were more active against dermatophyte.Compound (��7)��(��12 )��(��13)��(��15) showed equal or more activity compared with clotrimazole o r ketoconazole.Compound(��12)exhibited outstanding inhibitory activit y against Candida albicans and Cryptococcus neoformans.
, http://www.100md.com
Key words dioxolanes;triazole derivatives;antifungal activity
�������������ϸ��ɫ��P-450��ϣ��Ӷ����������ϸ��ɫ��P-450����ø������ë��C14��-�Ѽ�����Ӧ����������������ϳɣ���ë�������ʹ���������ֳ������������ȱ���������������������1���������Ϊ�����ҩ���о�������һ���ȵ�.��Ȼ�Ա�����60���ĩ���������������ʮ���ҩ�������ٴ������ɹ�������������ҩ���������Ҿ���һ���ľ�������2������ͪ�������ܿڷ���ҩ��������ʹ���ײ����ζ��ԣ���ˣ��ٴ�����Ҫ��Ч�Ͷ��������㡢ѡ���ԺõĿ�����ҩ��.
1 �ϳ�·��
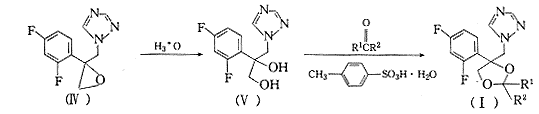
���ݵ��������Ŀ�������û�����Plempel��Wilkinson�Ե��������Ĺ�Ч��ϵ���о������3��4������Ʋ��ϳ���21���µ�2��2-˫ȡ��-4-(2��4-��������)-4-(1H-1��2��4-����-1-��)��-1��3-�����컷������ϳ�·��ͼ1.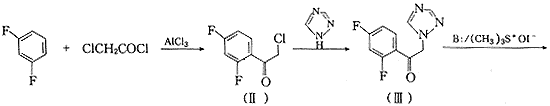
, http://www.100md.com
Fig.1 General synthetic route of the title compounds
2 ʵ�鲿��
�¶�δ��У�����۵���ZMD-1�͵����۵�ⶨ�Dzⶨ.�˴Ź�������Bruker Spectrospin A C-P 300���Dzⶨ��CDCl3Ϊ�ܼ���TMSΪ�ڱ�.
2.1 �ϳ�ʵ��
2-��-2�䣬4��-��������ͪ(��)��2�䣬4��-����-2-(1H-1��2��4-����-1-��)����ͪ(��)�������ס�5�������ϳɣ�2-(2��4-��������)-3-(1H-1��2��4-����-1-��)-1��2-������(��)�����ס�8���ϳ�.
2.1.1 2��2-˫ȡ��-4-(2��4-��������)-4-(1H-1��2��4-����-1-��)��-1��3-�����컷������(��)�ĺϳ�
, ��Ĵҽҩ
ȡ1.275g(5.0mmol)������2-(2��4-��������)-3-(1H-1��2��4-����-1-��)-1��2-��������5.0mmolȩ��ͪ��2.28g(6.0mmol)�Լױ�����һˮ�����60mL�ױ������Ȼ�������24h�����ϳ�ȥ��Ӧ�в�����ˮ����Ӧ�ϣ���ӦҺ�м��뱥��̼������ˮ��Һ�����ԣ���Һ������������ȡ����ˮ�����Ƹ����ҹ���������������������ᆳ�轺���������룬����������-������(1��1)Ϊϴ�Ѽ�����ϴ��������Ϊ˳ʽ�칹�壬��ϴ��������Ϊ��ʽ�칹����6��.
2.2 �����������
��ѡ��6���ٴ��ϳ������²����(Ϊ����ҽԺƤ��������ұ��صı�����)�������������������������������˿��������֦��ù����Ѭ��ɫ��ù������ɫëѢ��.
��������7���������Թ�����ɳ��(Sabouraud��s)�������ⶨ�����������־�Ũ��(MIC).�Ƚ���������������N��N-������������Ȼ����������ϡ����80mg/L���ٶԱ�ϡ����Ũ�ȣ�ͬʱ����ý���պͿհ��գ����ֺ���(29��1)������������������Ͱ����������2d��������������7d�������MICΪ����չ���Ƚ�û����������Ļ�����Ũ��.
, ��Ĵҽҩ
3 ���������
�ϳɵĻ���������˴Ź�������Ԫ�ط���ȷ֤�ṹ��������������������������ݼ���1.ͨ���Ա�ϡ�ͷ���6�ֳ����²�������������⿹����������飬���������������21��Ŀ�껯������в�ͬ�̶ȵĿ�������ԣ�һ��أ���dz���²�������־�Ч��Ҫ�ȶ������ã����л�����(��7)��(��12)��(��13)��(��15)�Ļ������ù���ͪ�����൱��������(��12)�������������������к�ǿ����������.��������2.
Tab.1 Structure,physical properties and spectral d ata of the title compounds
Compd.
R1
, ��Ĵҽҩ
R2
Conf.
Yield/%
mp/��
1H-NMR(CDCl3)��
��1
H
C6H5
cis
39.07
81��83
, http://www.100md.com
4.12��4.15(d,J=9.4Hz��1H)��4.53��4.57(d,J=14.4Hz��1H)��4.60��4.65(d,J=14.6Hz��1H)��4.83��4.87(dd,J=9.4��3.0Hz��1H)��5.80(s,1H)��6.81��6.94(m,2H)��7.44��7.51(m,6H),7.84
(s,1H)��7.98(s,1H)
��2
H
C6H5
trans
39.45
78��80
, ��Ĵҽҩ
4.38��4.41(dd,J=9.1��1.6Hz��1H)��4.43��4.46(dd,J=9.1,1.4
Hz��1H)��4.54��4.59(d,J=14.5Hz��1H)��4.72��4.77(d,J=14.5
Hz��1H)��5.79(s,1H)��6.81��6.86(m,2H)��7.35��7.44
(m,6H),7.86(s,1H)��8.14(s,1H)
(to be continued)
(Continued Tab.1)
Compd.
R1
, http://www.100md.com
R2
Conf.
Yield/%
mp/��
1H-NMR(CDCl3)��
��3
H
4-CH3OC6H4
trans
30.16
oil
, http://www.100md.com
3.81(s,1H)��4.41��4.42(m,2H)��4.53��4.58(d,J=14.7Hz��1H),4.71��4.75(d,J=14.3Hz��1H)��5.71(s,1H)��6.79��6.90
(m,4H)��7.30��7.42(m,3H)��7.85(s,1H)��8.13(s,1H)
��4
H
2-CH3OC6H4
cis
43.97
125��127
, ��Ĵҽҩ 3.88(s,1H)��4.09��4.12(dd,J=1.9��9.3Hz��1H)��4.52��
4.57(d,J=14.3Hz��1H)��4.64��4.69(d,J=14.1Hz��1H)��4.77��4.81(dd,J=3.0��9.3Hz��1H)��6.12(s,1H)��6.86��
7.07(m,4H)��7.38��7.53(m,3H),7.83(s,1H)��7.99(s,1H)
��5
H
2-CH3OC6H5
trans
30.07
, http://www.100md.com
96��98
3.88(s,3H)��4.34��4.37(dd,J=1.9��9.1Hz��1H)��4.39��
4.42(dd,J=1.6��9.1Hz��1H)��4.57��4.62(d,J=15.0Hz��1H)
��4.75��4.80(d,J=14.8Hz��1H)��5.28(s,1H)��6.77��6.95
(m,4H)��7.31��7.38(m,3H),7.82(s,1H)��8.21(s,1H)
��6
H
4-NO2C6H4
, http://www.100md.com
cis
34.54
133��134
4.16��4.20(dd,J=1.9��9.5Hz��1H)��4.48��4.53(d,J=14.5
Hz��1H)��4.59��4.64(d,J=14.4Hz��1H)��4.90��4.94(dd,J=
3.2��9.5Hz��1H)��5.87(s,1H)��6.90��6.97(m,2H)��7.51��
7.60(m,3H),7.83(s,1H)��7.86(s,1H)��8.28��8.32(m,2H)
��7
, ��Ĵҽҩ H
3-BrC6H4
cis
32.70
oil
4.12��4.16(dd,J=1.9��9.4Hz��1H)��4.51��4.55(d,J=14.4
Hz��1H)��4.58��4.62(d,J=14.4Hz��1H)��4.83��4.87(dd,J=
1.1��9.4Hz��1H)��5.75(s,1H)��6.87��6.94(m,2H)��7.31��
7.33(m,2H),7.46��7.51(m,1H)��7.56��7.59(m,2H)��7.84
, ��Ĵҽҩ
(s,1H)��7.91(s,1H)
��8
H
3-BrC6H4
trans
34.12
oil
4.32��4.36(dd,J=1.9��9.3Hz��2H)��4.46��4.50(dd,J=1.7��9.3Hz��1H)��4.53��4.58(d,J=14.6Hz��1H)��4.71��4.75
(d,J=14.6Hz��1H)��5.77(s,1H)��6.80��6.89(m,2H)��7.21��
, http://www.100md.com
7.38(m,3H),7.48��7.55(m,2H)��7.87(s,1H)��8.11(s,1H)
��9
H
3-ClC6H4
trans
40.68
oil
4.33��4.36(dd,J=1.9��9.3Hz��1H)��4.46��4.50(dd,J=1.7��9.5Hz��1H)��4.53��4.58(d,J=14.6Hz��1H)��4.71��4.76
(d,J=14.6Hz��1H)��5.78(s,1H)��6.80��6.90(m,2H)��7.28��
, ��Ĵҽҩ
7.38(m,5H),7.87(s,1H)��8.11(s,1H)
��10
H
2-furanyl
cis
43.24
83��85
4.07��4.11(dd,J=1.5��9.4Hz��1H)��4.59��4.64(d,J=14.3
Hz��1H)��4.71��4.75(d,J=14.3Hz��1H)��4.75��4.79(dd,J=
3.1��9.4Hz��1H)��5.93(s,1H)��6.44��6.45(m,1H)��6.54��
, ��Ĵҽҩ
6.65(m,1H),6.81��6.92(m,2H)��7.31��7.39(m,1H)��7.50��
7.54(m,1H)��7.76(s,1H)��8.14(s,1H)
��11
H
2-furanyl
trans
29.82
113��115
4.30��4.33(dd,J=1.5��9.2Hz��1H)��4.49��4.53(dd,J=2.4��9.2Hz��1H)��4.51��4.55(d,J=14.4Hz��1H)��4.66��4.71
, http://www.100md.com
(d,J=14.4Hz��1H)��5.99(s,1H)��6.30��6.34(m,2H)��6.82��
6.90(m,2H),7.37��7.40(m,1H)��7.42��7.50(m,1H)��7.86
(s,1H)��8.10(s,1H)
��12
H
C6H5CH2
cis
36.02
128��130
, ��Ĵҽҩ
4.02��4.06(dd,J=1.5��9.4Hz��1H)��4.14��4.15(d,J=3.0
Hz��2H)��4.52��4.57(d,J=14.3Hz��1H)��4.65��4.70(d,J=
14.3Hz��1H)��4.69��4.74(dd,J=3.4��9.4Hz��1H)��5.32��
5.34(t,J=3.0Hz,1H)��6.84��7.04(m,5H),7.30��7.37(m,3H)
��7.79(s,1H)��8.16(s,1H)
��13
H
C6H5CHCH
, http://www.100md.com
cis
33.06
oil
3.98��4.02(dd,J=1.7��9.4Hz��1H)��4.45��4.50(d,J=14.3
Hz��1H)��4.63��4.58(d,J=14.3Hz��1H)��4.73��4.77(dd,J=
3.3��9.3Hz��1H)��5.39��5.41(d,J=6.5Hz��1H)��6.01��
6.09(dd,J=6.5��16.0Hz��1H)��6.77��6.83(d,J=16.0Hz��1H)��6.86��6.92(m,1H)��7.33��7.51(m,7H)��7.87(s,1H)��8.16(s,1H)
, ��Ĵҽҩ ��14
H
C6H5CHCH
trans
29.81
93��95
4.29��4.33(dd,J=1.6��9.1Hz��1H)��4.35��4.39
(dd,J=1.3��9.2Hz��1H)��4.51��4.55(d,J=14.6Hz��1H)��4.67��4.72(d,J=14.5Hz��1H)��5.46��5.48(d,J=6.4Hz��1H)��6.05��6.12(dd,J=6.4��16.0Hz��1H)��6.72��6.78
, http://www.100md.com
(d,J=16.0Hz��1H)��6.84��6.90(m,2H)��7.31��7.53(m,6H)��7.86(s,1H)��8.12(s,1H)
(to be continued)
(Continued Tab.1)
Compd.
R1
R2
Conf.
Yield/%
mp/��
1H-NMR(CDCl3)��
, ��Ĵҽҩ
��15
H
n-C6H13
cis
32.41
oil
0.89��0.92(t,J=5.0Hz��3H)��1.23��1.45(m,8H)��1.65��1.74
(m,2H)��3.87��3.91(dd,J=1.8��9.4Hz��1H)��4.43��4.48
(d,J=14.3Hz,1H),4.50��4.55(d,J=14.3Hz,1H),4.61��4.65
, ��Ĵҽҩ
(dd,J=3.3��9.3Hz,1H)��4.84��4.88(t,J=5.0Hz,1H)��6.81��
6.90(m,2H)��7.33��7.41(m,1H)��7.80(s,1H)��8.14(s,1H)
��16
H
n-C6H13
trans
31.20
oil
0.86��0.92(t,J=6.3Hz��3H)��1.22��1.42(m,8H)��1.61��1.72
, ��Ĵҽҩ
(m,2H)��4.20��4.24(m,2H)��4.47��4.52(d,J=14.0
Hz,1H),4.62��4.67(d,J=14.2Hz,1H),4.88��4.91(t,J=5.2
Hz,1H)��6.80��6.88(m,2H)��7.37��7.45(m,1H)��7.81(s,1H)��8.08(s,1H)
��17
��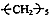
95.22
79��81
, ��Ĵҽҩ
1.42��1.71(m,10H)��4.18��4.24(dd,J=1.7��9.5Hz��1H)��4.43��4.48(d,J=14.4Hz��1H)��4.49��4.54
(d,J=14.3Hz��1H)��4.55��4.59(dd,J=3.2��9.4Hz,1H),6.78��6.88(m,2H)��7.35��7.46(m,1H)��7.80(s,1H)��8.11(s,1H)
��18
��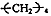
93.46
84��85
, ��Ĵҽҩ
1.59��1.89(m,8H)��3.99��4.03(dd,J=1.6��9.4Hz��1H)��4.39��4.44(d,J=14.2Hz��1H)��4.51��4.56(d,J=14.2Hz,1H),4.54��4.58(dd��J=3.3��9.4Hz��1H)��6.81��6.89(m,2H)��7.30��7.44(m,1H)��7.82(s,1H),8.09(s,1H)
��19
CH3
C6H5
cis
34.56
90��92
1.66(s,3H)��4.23��4.28(d,J=14.3Hz��2H)��4.37��4.41(dd,J=1.8��9.4Hz��1H)��4.41��4.45(dd,J=1.8��9.3Hz��1H)��4.48��4.53(d,J=14.3Hz��1H)��6.79��6.85(m,2H)��7.37��7.46(m,4H)��7.57��7.60(m,2H)��7.63(s,1H),7.75(s,1H)
, ��Ĵҽҩ
��20
CH3
C6H5
trans
34.17
102��104
1.69(s,3H)��3.87��3.91(dd,J=1.4��9.5Hz��1H)��4.45��4.52(d,J=14.2Hz��1H)��4.54��4.59(d,J=14.2Hz��1H)��4.72��4.77(dd,J=4.7��9.5Hz��1H)��6.69��6.75(m,2H)��7.20��7.41(m,6H)��7.85(s,1H)��8.13(s,1H)
��21
, ��Ĵҽҩ
CH3
4-ClC6H4
trans
42.96
133��134
1.66(s,3H)��3.84��3.88(dd,J=1.5��9.6Hz��1H)��4.45��4.50(d,J=14.2Hz��1H)��4.52��4.57
(d,J=14.2Hz��1H)��4.73��4.78(dd,J=4.6��9.6Hz��1H)��6.71��6.77(m,2H)��7.20��7.35(m,5H)��7.86(s,1H)��8.10(s,1H)
��C��H��N analyses were within��0.5% of calculated values ��Recrystalizing solvent��AcOEt-cyclohexane(1��1)Tab.2 Antifungal activities of the title compounds
, http://www.100md.com
MIC/mg.L-1
Compd.
Candida
albicans
Cryptococcus
neoforms
Sporotrichum
sch enckii
Cladosporium
carrionii
Aspergillus
, ��Ĵҽҩ
fumigatus
Trichophyton
rubum
��1
40
20
>80
20
>80
2.5
��2
20
, http://www.100md.com 40
>80
10
>80
1.25
��3
40
5
40
10
80
20
��4
, http://www.100md.com
>80
20
>80
80
80
10
��5
40
40
80
80
>80
80
, http://www.100md.com
��6
>80
80
80
20
80
5
��7
10
5
2.5
2.5
40
, http://www.100md.com
1.25
��8
5
40
40
10
40
1.25
��9
20
5
20
20
, http://www.100md.com
40
5
��10
80
>80
>80
5
>80
2.5
��11
>80
40
40
, ��Ĵҽҩ
20
80
2.5
��12
0.625
0.156
20
5
20
0.312
(to be continued)
(Continued Tab.2)
, ��Ĵҽҩ
Compd.
Candida
albicans
Cryptococcus
neoformans
Sporotrichum
schenckii
Cladosporium
carrionii
Aspergillus
fumigatus
, ��Ĵҽҩ Trichophyton
rubum
��13
5
20
40
2.5
5
1.25
��14
5
20
40
, ��Ĵҽҩ
5
40
2.5
��15
5
5
20
10
20
2.5
��16
20
5
, ��Ĵҽҩ
40
10
40
2.5
��17
40
40
40
10
20
2.5
��18
80
, ��Ĵҽҩ
80
80
20
40
2.5
��19
40
20
40
5
40
2.5
��20
, ��Ĵҽҩ
10
20
20
40
80
20
��21
80
80
40
80
80
2.5
, ��Ĵҽҩ
clotrimazole
10
2.5
20
1.25
10
0.156
ketoconazole
5
1.25
10
1.25
10
, ��Ĵҽҩ
0.625
2-��������±ԭ��ȡ��ʱ�����������������ǿ��˳ʽ���Ϳ���������뷴ʽ���Ͳ������.
1�ܺ������ѧ������������No.Y-8
����ܣ�ͨѶ��ϵ��
�����
1 Yoshida Y,Aoyama Y.Interaction of azole antifungal agents with cytoch rome P-450DM purified from Saccharomyces cerevisiae microsome.Biochem P harmacol,1987��36(2)��229��235
2 ����.ȫ���ÿ�ù����.����ҩѧ�鱨��1991��10(1)��3��5
, http://www.100md.com
3 Plempel M.Experience,recognitions and questions in azole antimycotics.Jpn Med Mycol��1982��23(1)��17��23
4 Wilkinson CF,Krystyna H.Structure-activity relationships in the effects of 1-alkylimidazoles on microsomal oxidation in vitro and in vivo.Biochem Pharmacol,1974��23(17)��2377��2381
5 Richardson K.Triazoles.Brit UK Pat Appl,Cl.C07D249/08.GB 2099818A.1982-12-1 5
6 Saksena AK,Cooper AB,Guzik H��et al.Preparation and testing of 2��4��4-t r i-and 2��2��4��4-tetra substituted-1��3-dioxolanes as drugs.PTC Int Appl.Cl. C07D405/06��WO 88/05048.1988-07-14
7 Stutz A,Georgopoulos A,Granitzer W��et al.Synthesis and structure-activ it y relationships of naftifine-related allyamine antimycotics.J Med Chem,1986��29 (1)��112��125
8 ����ܣ������Ŵ�־����.���������ĺϳɼ��俹�������(��).�й�ҩ�ﻯѧ��־��1998��8(4)��235��238
�ո�����:1999-04-27, ��Ĵҽҩ
��λ���ڶ���ҽ��ѧҩѧԺ���Ϻ�200433
�ؼ��ʣ������컷����������������������
ժ Ҫ Ϊ��Ѱ������Ч�Ͷ��Ŀ����ҩժ Ҫ Ϊ��Ѱ������Ч�Ͷ��Ŀ����ҩ�����ݵ�������ҩ��Ĺ�Ч��ϵ�����û�������ƺϳ���21��2��2-˫ȡ��-4-(2��4-��������)-4-(1H-1��2��4-����-1-��)��-1��3�����컷������ṹ��Ԫ�ط�������˴Ź�����֤ʵ.���������⿹�������������������е�Ŀ�껯�����6�ֳ������²�������в�ͬ�̶ȵĿ�������ԣ���dz�������Ч��Ҫ�ȶ�������Ч���ã����л�����(��7)��(��12)��(��13)��(��15)�Ļ������ù���ͪ�����൱��������(��12)�������������������к�ǿ����������.
, http://www.100md.com
Synthesis and Antifungal Activity of Triazole Derivatives ��
Wu Yijie,Yang Jiqiu,Zhou Tingsen,Zhang Dazhi,Liu Chaomei,Zhou Mingde
(College of Pharmacy,Second Military Medical University,Shanghai 200433 )
Abstract In an effort to search for more potent and less toxi c antifungal agents,according to the structure-activity relationships and antin ycotic mechanism of the azole compounds,we designed and synthesized twenty-one 2��2-disu bstituted-4-(2��4-difluorophenyl)-4-(1H-1��2��4-triazol-1-yl)methyl -1,3-dioxolane.Their structures were confirmed by 1H-NMR and elementar y analysis.Preliminary results of the antifungal tests in vitro showed that all compoun d s exhibited activity against the common pathogenic fungi.Generally,the title com pounds were more active against dermatophyte.Compound (��7)��(��12 )��(��13)��(��15) showed equal or more activity compared with clotrimazole o r ketoconazole.Compound(��12)exhibited outstanding inhibitory activit y against Candida albicans and Cryptococcus neoformans.
, http://www.100md.com
Key words dioxolanes;triazole derivatives;antifungal activity
�������������ϸ��ɫ��P-450��ϣ��Ӷ����������ϸ��ɫ��P-450����ø������ë��C14��-�Ѽ�����Ӧ����������������ϳɣ���ë�������ʹ���������ֳ������������ȱ���������������������1���������Ϊ�����ҩ���о�������һ���ȵ�.��Ȼ�Ա�����60���ĩ���������������ʮ���ҩ�������ٴ������ɹ�������������ҩ���������Ҿ���һ���ľ�������2������ͪ�������ܿڷ���ҩ��������ʹ���ײ����ζ��ԣ���ˣ��ٴ�����Ҫ��Ч�Ͷ��������㡢ѡ���ԺõĿ�����ҩ��.
1 �ϳ�·��
���ݵ��������Ŀ�������û�����Plempel��Wilkinson�Ե��������Ĺ�Ч��ϵ���о������3��4������Ʋ��ϳ���21���µ�2��2-˫ȡ��-4-(2��4-��������)-4-(1H-1��2��4-����-1-��)��-1��3-�����컷������ϳ�·��ͼ1.


, http://www.100md.com
Fig.1 General synthetic route of the title compounds
2 ʵ�鲿��
�¶�δ��У�����۵���ZMD-1�͵����۵�ⶨ�Dzⶨ.�˴Ź�������Bruker Spectrospin A C-P 300���Dzⶨ��CDCl3Ϊ�ܼ���TMSΪ�ڱ�.
2.1 �ϳ�ʵ��
2-��-2�䣬4��-��������ͪ(��)��2�䣬4��-����-2-(1H-1��2��4-����-1-��)����ͪ(��)�������ס�5�������ϳɣ�2-(2��4-��������)-3-(1H-1��2��4-����-1-��)-1��2-������(��)�����ס�8���ϳ�.
2.1.1 2��2-˫ȡ��-4-(2��4-��������)-4-(1H-1��2��4-����-1-��)��-1��3-�����컷������(��)�ĺϳ�
, ��Ĵҽҩ
ȡ1.275g(5.0mmol)������2-(2��4-��������)-3-(1H-1��2��4-����-1-��)-1��2-��������5.0mmolȩ��ͪ��2.28g(6.0mmol)�Լױ�����һˮ�����60mL�ױ������Ȼ�������24h�����ϳ�ȥ��Ӧ�в�����ˮ����Ӧ�ϣ���ӦҺ�м��뱥��̼������ˮ��Һ�����ԣ���Һ������������ȡ����ˮ�����Ƹ����ҹ���������������������ᆳ�轺���������룬����������-������(1��1)Ϊϴ�Ѽ�����ϴ��������Ϊ˳ʽ�칹�壬��ϴ��������Ϊ��ʽ�칹����6��.
2.2 �����������
��ѡ��6���ٴ��ϳ������²����(Ϊ����ҽԺƤ��������ұ��صı�����)�������������������������������˿��������֦��ù����Ѭ��ɫ��ù������ɫëѢ��.
��������7���������Թ�����ɳ��(Sabouraud��s)�������ⶨ�����������־�Ũ��(MIC).�Ƚ���������������N��N-������������Ȼ����������ϡ����80mg/L���ٶԱ�ϡ����Ũ�ȣ�ͬʱ����ý���պͿհ��գ����ֺ���(29��1)������������������Ͱ����������2d��������������7d�������MICΪ����չ���Ƚ�û����������Ļ�����Ũ��.
, ��Ĵҽҩ
3 ���������
�ϳɵĻ���������˴Ź�������Ԫ�ط���ȷ֤�ṹ��������������������������ݼ���1.ͨ���Ա�ϡ�ͷ���6�ֳ����²�������������⿹����������飬���������������21��Ŀ�껯������в�ͬ�̶ȵĿ�������ԣ�һ��أ���dz���²�������־�Ч��Ҫ�ȶ������ã����л�����(��7)��(��12)��(��13)��(��15)�Ļ������ù���ͪ�����൱��������(��12)�������������������к�ǿ����������.��������2.
Tab.1 Structure,physical properties and spectral d ata of the title compounds
Compd.
R1
, ��Ĵҽҩ
R2
Conf.
Yield/%
mp/��
1H-NMR(CDCl3)��
��1
H
C6H5
cis
39.07
81��83
, http://www.100md.com
4.12��4.15(d,J=9.4Hz��1H)��4.53��4.57(d,J=14.4Hz��1H)��4.60��4.65(d,J=14.6Hz��1H)��4.83��4.87(dd,J=9.4��3.0Hz��1H)��5.80(s,1H)��6.81��6.94(m,2H)��7.44��7.51(m,6H),7.84
(s,1H)��7.98(s,1H)
��2
H
C6H5
trans
39.45
78��80
, ��Ĵҽҩ
4.38��4.41(dd,J=9.1��1.6Hz��1H)��4.43��4.46(dd,J=9.1,1.4
Hz��1H)��4.54��4.59(d,J=14.5Hz��1H)��4.72��4.77(d,J=14.5
Hz��1H)��5.79(s,1H)��6.81��6.86(m,2H)��7.35��7.44
(m,6H),7.86(s,1H)��8.14(s,1H)
(to be continued)
(Continued Tab.1)
Compd.
R1
, http://www.100md.com
R2
Conf.
Yield/%
mp/��
1H-NMR(CDCl3)��
��3
H
4-CH3OC6H4
trans
30.16
oil
, http://www.100md.com
3.81(s,1H)��4.41��4.42(m,2H)��4.53��4.58(d,J=14.7Hz��1H),4.71��4.75(d,J=14.3Hz��1H)��5.71(s,1H)��6.79��6.90
(m,4H)��7.30��7.42(m,3H)��7.85(s,1H)��8.13(s,1H)
��4
H
2-CH3OC6H4
cis
43.97
125��127
, ��Ĵҽҩ 3.88(s,1H)��4.09��4.12(dd,J=1.9��9.3Hz��1H)��4.52��
4.57(d,J=14.3Hz��1H)��4.64��4.69(d,J=14.1Hz��1H)��4.77��4.81(dd,J=3.0��9.3Hz��1H)��6.12(s,1H)��6.86��
7.07(m,4H)��7.38��7.53(m,3H),7.83(s,1H)��7.99(s,1H)
��5
H
2-CH3OC6H5
trans
30.07
, http://www.100md.com
96��98
3.88(s,3H)��4.34��4.37(dd,J=1.9��9.1Hz��1H)��4.39��
4.42(dd,J=1.6��9.1Hz��1H)��4.57��4.62(d,J=15.0Hz��1H)
��4.75��4.80(d,J=14.8Hz��1H)��5.28(s,1H)��6.77��6.95
(m,4H)��7.31��7.38(m,3H),7.82(s,1H)��8.21(s,1H)
��6
H
4-NO2C6H4
, http://www.100md.com
cis
34.54
133��134
4.16��4.20(dd,J=1.9��9.5Hz��1H)��4.48��4.53(d,J=14.5
Hz��1H)��4.59��4.64(d,J=14.4Hz��1H)��4.90��4.94(dd,J=
3.2��9.5Hz��1H)��5.87(s,1H)��6.90��6.97(m,2H)��7.51��
7.60(m,3H),7.83(s,1H)��7.86(s,1H)��8.28��8.32(m,2H)
��7
, ��Ĵҽҩ H
3-BrC6H4
cis
32.70
oil
4.12��4.16(dd,J=1.9��9.4Hz��1H)��4.51��4.55(d,J=14.4
Hz��1H)��4.58��4.62(d,J=14.4Hz��1H)��4.83��4.87(dd,J=
1.1��9.4Hz��1H)��5.75(s,1H)��6.87��6.94(m,2H)��7.31��
7.33(m,2H),7.46��7.51(m,1H)��7.56��7.59(m,2H)��7.84
, ��Ĵҽҩ
(s,1H)��7.91(s,1H)
��8
H
3-BrC6H4
trans
34.12
oil
4.32��4.36(dd,J=1.9��9.3Hz��2H)��4.46��4.50(dd,J=1.7��9.3Hz��1H)��4.53��4.58(d,J=14.6Hz��1H)��4.71��4.75
(d,J=14.6Hz��1H)��5.77(s,1H)��6.80��6.89(m,2H)��7.21��
, http://www.100md.com
7.38(m,3H),7.48��7.55(m,2H)��7.87(s,1H)��8.11(s,1H)
��9
H
3-ClC6H4
trans
40.68
oil
4.33��4.36(dd,J=1.9��9.3Hz��1H)��4.46��4.50(dd,J=1.7��9.5Hz��1H)��4.53��4.58(d,J=14.6Hz��1H)��4.71��4.76
(d,J=14.6Hz��1H)��5.78(s,1H)��6.80��6.90(m,2H)��7.28��
, ��Ĵҽҩ
7.38(m,5H),7.87(s,1H)��8.11(s,1H)
��10
H
2-furanyl
cis
43.24
83��85
4.07��4.11(dd,J=1.5��9.4Hz��1H)��4.59��4.64(d,J=14.3
Hz��1H)��4.71��4.75(d,J=14.3Hz��1H)��4.75��4.79(dd,J=
3.1��9.4Hz��1H)��5.93(s,1H)��6.44��6.45(m,1H)��6.54��
, ��Ĵҽҩ
6.65(m,1H),6.81��6.92(m,2H)��7.31��7.39(m,1H)��7.50��
7.54(m,1H)��7.76(s,1H)��8.14(s,1H)
��11
H
2-furanyl
trans
29.82
113��115
4.30��4.33(dd,J=1.5��9.2Hz��1H)��4.49��4.53(dd,J=2.4��9.2Hz��1H)��4.51��4.55(d,J=14.4Hz��1H)��4.66��4.71
, http://www.100md.com
(d,J=14.4Hz��1H)��5.99(s,1H)��6.30��6.34(m,2H)��6.82��
6.90(m,2H),7.37��7.40(m,1H)��7.42��7.50(m,1H)��7.86
(s,1H)��8.10(s,1H)
��12
H
C6H5CH2
cis
36.02
128��130
, ��Ĵҽҩ
4.02��4.06(dd,J=1.5��9.4Hz��1H)��4.14��4.15(d,J=3.0
Hz��2H)��4.52��4.57(d,J=14.3Hz��1H)��4.65��4.70(d,J=
14.3Hz��1H)��4.69��4.74(dd,J=3.4��9.4Hz��1H)��5.32��
5.34(t,J=3.0Hz,1H)��6.84��7.04(m,5H),7.30��7.37(m,3H)
��7.79(s,1H)��8.16(s,1H)
��13
H
C6H5CHCH
, http://www.100md.com
cis
33.06
oil
3.98��4.02(dd,J=1.7��9.4Hz��1H)��4.45��4.50(d,J=14.3
Hz��1H)��4.63��4.58(d,J=14.3Hz��1H)��4.73��4.77(dd,J=
3.3��9.3Hz��1H)��5.39��5.41(d,J=6.5Hz��1H)��6.01��
6.09(dd,J=6.5��16.0Hz��1H)��6.77��6.83(d,J=16.0Hz��1H)��6.86��6.92(m,1H)��7.33��7.51(m,7H)��7.87(s,1H)��8.16(s,1H)
, ��Ĵҽҩ ��14
H
C6H5CHCH
trans
29.81
93��95
4.29��4.33(dd,J=1.6��9.1Hz��1H)��4.35��4.39
(dd,J=1.3��9.2Hz��1H)��4.51��4.55(d,J=14.6Hz��1H)��4.67��4.72(d,J=14.5Hz��1H)��5.46��5.48(d,J=6.4Hz��1H)��6.05��6.12(dd,J=6.4��16.0Hz��1H)��6.72��6.78
, http://www.100md.com
(d,J=16.0Hz��1H)��6.84��6.90(m,2H)��7.31��7.53(m,6H)��7.86(s,1H)��8.12(s,1H)
(to be continued)
(Continued Tab.1)
Compd.
R1
R2
Conf.
Yield/%
mp/��
1H-NMR(CDCl3)��
, ��Ĵҽҩ
��15
H
n-C6H13
cis
32.41
oil
0.89��0.92(t,J=5.0Hz��3H)��1.23��1.45(m,8H)��1.65��1.74
(m,2H)��3.87��3.91(dd,J=1.8��9.4Hz��1H)��4.43��4.48
(d,J=14.3Hz,1H),4.50��4.55(d,J=14.3Hz,1H),4.61��4.65
, ��Ĵҽҩ
(dd,J=3.3��9.3Hz,1H)��4.84��4.88(t,J=5.0Hz,1H)��6.81��
6.90(m,2H)��7.33��7.41(m,1H)��7.80(s,1H)��8.14(s,1H)
��16
H
n-C6H13
trans
31.20
oil
0.86��0.92(t,J=6.3Hz��3H)��1.22��1.42(m,8H)��1.61��1.72
, ��Ĵҽҩ
(m,2H)��4.20��4.24(m,2H)��4.47��4.52(d,J=14.0
Hz,1H),4.62��4.67(d,J=14.2Hz,1H),4.88��4.91(t,J=5.2
Hz,1H)��6.80��6.88(m,2H)��7.37��7.45(m,1H)��7.81(s,1H)��8.08(s,1H)
��17
��
95.22
79��81
, ��Ĵҽҩ
1.42��1.71(m,10H)��4.18��4.24(dd,J=1.7��9.5Hz��1H)��4.43��4.48(d,J=14.4Hz��1H)��4.49��4.54
(d,J=14.3Hz��1H)��4.55��4.59(dd,J=3.2��9.4Hz,1H),6.78��6.88(m,2H)��7.35��7.46(m,1H)��7.80(s,1H)��8.11(s,1H)
��18
��
93.46
84��85
, ��Ĵҽҩ
1.59��1.89(m,8H)��3.99��4.03(dd,J=1.6��9.4Hz��1H)��4.39��4.44(d,J=14.2Hz��1H)��4.51��4.56(d,J=14.2Hz,1H),4.54��4.58(dd��J=3.3��9.4Hz��1H)��6.81��6.89(m,2H)��7.30��7.44(m,1H)��7.82(s,1H),8.09(s,1H)
��19
CH3
C6H5
cis
34.56
90��92
1.66(s,3H)��4.23��4.28(d,J=14.3Hz��2H)��4.37��4.41(dd,J=1.8��9.4Hz��1H)��4.41��4.45(dd,J=1.8��9.3Hz��1H)��4.48��4.53(d,J=14.3Hz��1H)��6.79��6.85(m,2H)��7.37��7.46(m,4H)��7.57��7.60(m,2H)��7.63(s,1H),7.75(s,1H)
, ��Ĵҽҩ
��20
CH3
C6H5
trans
34.17
102��104
1.69(s,3H)��3.87��3.91(dd,J=1.4��9.5Hz��1H)��4.45��4.52(d,J=14.2Hz��1H)��4.54��4.59(d,J=14.2Hz��1H)��4.72��4.77(dd,J=4.7��9.5Hz��1H)��6.69��6.75(m,2H)��7.20��7.41(m,6H)��7.85(s,1H)��8.13(s,1H)
��21
, ��Ĵҽҩ
CH3
4-ClC6H4
trans
42.96
133��134
1.66(s,3H)��3.84��3.88(dd,J=1.5��9.6Hz��1H)��4.45��4.50(d,J=14.2Hz��1H)��4.52��4.57
(d,J=14.2Hz��1H)��4.73��4.78(dd,J=4.6��9.6Hz��1H)��6.71��6.77(m,2H)��7.20��7.35(m,5H)��7.86(s,1H)��8.10(s,1H)
��C��H��N analyses were within��0.5% of calculated values ��Recrystalizing solvent��AcOEt-cyclohexane(1��1)Tab.2 Antifungal activities of the title compounds
, http://www.100md.com
MIC/mg.L-1
Compd.
Candida
albicans
Cryptococcus
neoforms
Sporotrichum
sch enckii
Cladosporium
carrionii
Aspergillus
, ��Ĵҽҩ
fumigatus
Trichophyton
rubum
��1
40
20
>80
20
>80
2.5
��2
20
, http://www.100md.com 40
>80
10
>80
1.25
��3
40
5
40
10
80
20
��4
, http://www.100md.com
>80
20
>80
80
80
10
��5
40
40
80
80
>80
80
, http://www.100md.com
��6
>80
80
80
20
80
5
��7
10
5
2.5
2.5
40
, http://www.100md.com
1.25
��8
5
40
40
10
40
1.25
��9
20
5
20
20
, http://www.100md.com
40
5
��10
80
>80
>80
5
>80
2.5
��11
>80
40
40
, ��Ĵҽҩ
20
80
2.5
��12
0.625
0.156
20
5
20
0.312
(to be continued)
(Continued Tab.2)
, ��Ĵҽҩ
Compd.
Candida
albicans
Cryptococcus
neoformans
Sporotrichum
schenckii
Cladosporium
carrionii
Aspergillus
fumigatus
, ��Ĵҽҩ Trichophyton
rubum
��13
5
20
40
2.5
5
1.25
��14
5
20
40
, ��Ĵҽҩ
5
40
2.5
��15
5
5
20
10
20
2.5
��16
20
5
, ��Ĵҽҩ
40
10
40
2.5
��17
40
40
40
10
20
2.5
��18
80
, ��Ĵҽҩ
80
80
20
40
2.5
��19
40
20
40
5
40
2.5
��20
, ��Ĵҽҩ
10
20
20
40
80
20
��21
80
80
40
80
80
2.5
, ��Ĵҽҩ
clotrimazole
10
2.5
20
1.25
10
0.156
ketoconazole
5
1.25
10
1.25
10
, ��Ĵҽҩ
0.625
2-��������±ԭ��ȡ��ʱ�����������������ǿ��˳ʽ���Ϳ���������뷴ʽ���Ͳ������.
1�ܺ������ѧ������������No.Y-8
����ܣ�ͨѶ��ϵ��
�����
1 Yoshida Y,Aoyama Y.Interaction of azole antifungal agents with cytoch rome P-450DM purified from Saccharomyces cerevisiae microsome.Biochem P harmacol,1987��36(2)��229��235
2 ����.ȫ���ÿ�ù����.����ҩѧ�鱨��1991��10(1)��3��5
, http://www.100md.com
3 Plempel M.Experience,recognitions and questions in azole antimycotics.Jpn Med Mycol��1982��23(1)��17��23
4 Wilkinson CF,Krystyna H.Structure-activity relationships in the effects of 1-alkylimidazoles on microsomal oxidation in vitro and in vivo.Biochem Pharmacol,1974��23(17)��2377��2381
5 Richardson K.Triazoles.Brit UK Pat Appl,Cl.C07D249/08.GB 2099818A.1982-12-1 5
6 Saksena AK,Cooper AB,Guzik H��et al.Preparation and testing of 2��4��4-t r i-and 2��2��4��4-tetra substituted-1��3-dioxolanes as drugs.PTC Int Appl.Cl. C07D405/06��WO 88/05048.1988-07-14
7 Stutz A,Georgopoulos A,Granitzer W��et al.Synthesis and structure-activ it y relationships of naftifine-related allyamine antimycotics.J Med Chem,1986��29 (1)��112��125
8 ����ܣ������Ŵ�־����.���������ĺϳɼ��俹�������(��).�й�ҩ�ﻯѧ��־��1998��8(4)��235��238
�ո�����:1999-04-27, ��Ĵҽҩ